This article is mainly about the related introduction of lmp91000, and focuses on the application of lmp91000 in electrochemical sensor electrode fault detection.
Electrochemical sensors1. Electrochemical gas sensor
Electrochemical sensors operate by reacting with analytes and generating electrical signals. Most electrochemical gas sensors are current sensors, which generate a current that is linearly proportional to the gas concentration. The principle of the current measuring sensor is to measure the current-potential relationship in an electrochemical cell that has not established a balance. The current is quantitatively related to the rate of the electrolysis process of the sensing electrode (also called the working electrode), which is usually maintained at a constant potential using another electrode (the so-called reference electrode)
2. Working principle
The working principle of an electrochemical gas sensor is as follows: the target gas molecules in contact with the sensor first pass through a membrane to prevent condensation, which also acts as a dustproof. Then the gas molecules diffuse through the capillary, possibly through the subsequent filter, and then through the hydrophobic membrane to reach the surface of the sensing electrode. There, the molecules are immediately oxidized or reduced, thereby generating or consuming electrons, thereby generating electric current.
It is important to note that the amount of gas molecules entering the sensor in this way is limited by diffusion through the capillary. By optimizing the path, according to the desired measurement range, the appropriate electrical signal can be obtained. The design of the sensing electrode is essential to achieve high reactivity to the target gas and suppress the undesired response to the interfering gas. It involves a three-stage system of solids, liquids and gases, and all involve the chemical identification of analyte gases. Committed to tailoring the system and obtaining high-performance gas sensors. The electrochemical cell is completed by the so-called counter electrode, the Cont electrode, which balances the reaction at the sensing electrode. The ionic current between the Cont electrode and the Sen electrode is carried by the electrolyte in the sensor body, and the current path is provided by a wire terminated with a pin connector. The third electrode is usually included in an electrochemical sensor (3-electrode sensor). The so-called reference electrode is used to maintain the potential of the sensing electrode at a fixed value. For this and usually used for the operation of electrochemical sensors, a potentiostatic circuit is required.
3. Sensor signal
Sensor signal The output signal of a gas sensor corresponds to the concentration of the gas rather than its partial pressure. Therefore, one sensor can be used at different altitudes or even underground, regardless of the atmospheric pressure at which the device is calibrated. A more in-depth and scientific explanation of the correlation between sensor output and pressure can be found in the file mem4.
Application of lmp91000 in Electrochemical Sensor Electrode Fault Detection1 Electrochemical sensor and its signal conditioning circuit The working principle of the electrochemical gas sensor is similar to that of the galvanic cell. When the sensitive gas diffuses into the sensor, the oxidation-reduction reaction occurs, and the charge carrier and gas concentration output during the chemical reaction process Directly proportional. In most cases, three-electrode sensors are more widely used. Compared with early two-electrode gas sensors, three-electrode gas sensors have higher detection sensitivity, are easier to stabilize, and can achieve high-concentration detection. Figure -1 is a schematic diagram of the electrochemical sensor and its signal conditioning circuit. The sensor (SENSOR) part shows the electrode composition of the three-electrode chemical sensor and its equivalent circuit. The role of the operational amplifier in the circuit makes the reference electrode (RE ) Maintain a constant potential relative to the working electrode (WE), which collects the current generated by the sensitive gas in the reaction and realizes the amplification of the weak current signal through the post-op amplifier. The detection efficiency and sensitivity of the three-electrode sensor are obviously affected by the bias voltage; the selectivity of the sensor to sensitive gases will also be affected by the bias voltage; the bias voltage should be kept constant during the application process.
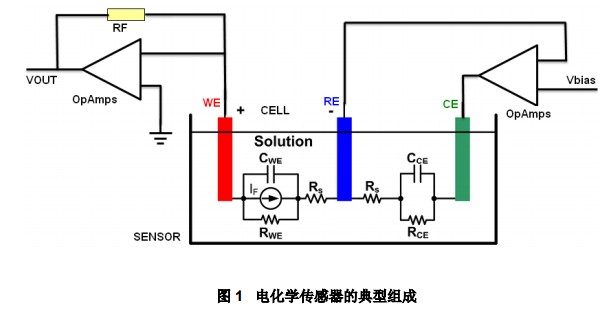
A common three-electrode electrochemical sensor signal conditioning circuit is composed of a bias circuit unit and a transimpedance amplifier unit; the former ensures the best working conditions of the sensor, and the latter realizes the amplification of the output weak current signal. In traditional design schemes, a dual-channel op amp is usually used to realize the design. Since the output of the electrochemical sensor is sensitive to the bias voltage, the op amp in the bias unit is required to have a lower offset voltage and smaller temperature drift characteristics, otherwise the stability of the sensor will be significantly affected; the implementation of the transimpedance amplifier For the amplification of the weak and small current signal output by the working electrode, it is hoped that the bias current of the op amp is small, otherwise the bias current is superimposed on the output current of the sensor to cause the output to have a large bias error. According to the above analysis, the characteristics of the selected dual op amp should simultaneously meet the low offset voltage, small bias current, and low power consumption. In many cases, the dual op amp models that meet the above conditions at the same time are very limited.
Due to the characteristics of electrochemical sensors, metal short-circuit caps are usually required to short-circuit the output after the sensor is manufactured to prevent charge accumulation, otherwise there will be a longer period of aging (stabilisation) after the sensor is put into use; for some sensors with zero bias voltage The short-circuit cap is also required to maintain a constant potential; for sensors with a non-zero bias voltage, the bias voltage is required to be maintained after the manufacturing is completed until the final field activation, otherwise a long-term aging process will also occur. In order to prevent sensor assembly errors and ensure that the characteristics of the sensor in use are not significantly degraded, it is necessary to monitor the electrode connection state of the sensor, which is also a necessary measure for safe use. Obviously, solutions based on traditional dual op amps are very challenging in design and commissioning when implementing such functions. Some fault detection solutions based on other discrete devices are often difficult to solve the problem of commissioning and device consistency.
There are two functional units, a bias voltage generator and a transimpedance amplifier, and the working parameters of each unit can be programmed and configured through the I2C bus, so that it can be flexibly adapted to the application of sensors with different characteristics. In order to facilitate the realization of temperature compensation, a semiconductor temperature sensor is integrated on the chip, and the temperature signal output can be time-sharing output or separate output. The internal functional block diagram of LMP91000 is shown in Figure-2 below. Compared with the traditional dual op amp solution, LMP91000 has obvious low power consumption, the average power consumption is only 10uA; PCB packaging area is small, only 5x5mm; high flexibility, programmable setting function unit required operating parameter points.
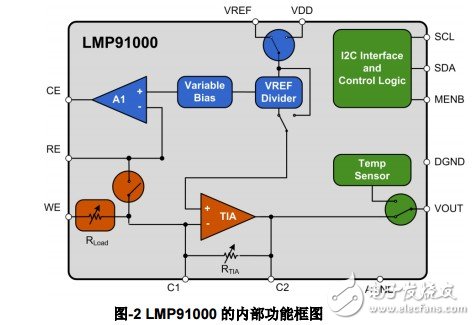
As mentioned above, the bias voltage in the application has a great influence on the working state of the electrochemical sensor. When the bias voltage changes slightly, the corresponding output current will change significantly. This phenomenon can be explained from the equivalent circuit of the electrochemical sensor. Due to the large equivalent capacitance between the electrodes, a small voltage change trend will produce a large current flow between the electrodes in an attempt to resist this change. If this bias voltage change is small and the duration is short, it will not affect the electrode, such as electrode polarization. This principle can usually be used to detect the connection status of the electrodes, especially the LMP91000 is relatively simple to implement. It only needs to dynamically configure the bias voltage of the sensor through the I2C bus to generate artificial disturbance of the bias voltage; it is monitored in real time in the process The change trend of the sensor voltage is sufficient.
2 Electrochemical sensor electrode failure detection
The internal working parameters of LMP91000 can be configured flexibly at any time, which can easily realize electrochemical sensor electrode fault detection. The following tests are performed on the LMP91000 evaluation board and the corresponding GUI software. The electrochemical sensor used is a two-electrode carbon monoxide sensor. In the experiment, the sensor function is normal, the sensor electrode is short-circuited, and the sensor is missing. The following is the description of the waveform and data characteristics of the sensor output voltage under various conditions.
2.1 Normal use of the sensor This test is to check whether the sensor function is normal. The following Figure-3 and Figure-4 respectively show the output voltage of the sensor before and after the Sensor Check function in the GUI is enabled. The following figure-3 shows the output condition of the sensor in normal application. It can be seen that the output signal is relatively smooth. Note that the Sensor Check function is not enabled at this time.
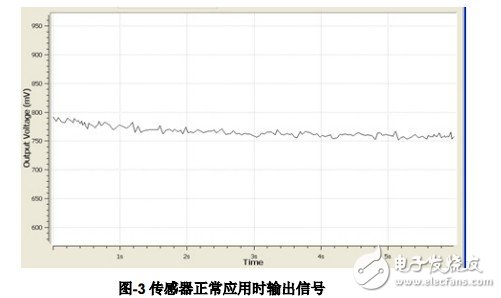
Figure-4 corresponds to the LMP91000 output after the Sensor Check function is activated. The output voltage drops from the original normal value and stays at a low level for a period of time, and then the output voltage climbs to the normal value. Observing the output voltage waveform carefully, you can find that the rebound voltage has a more obvious overshoot, which is about 300mV compared to the normal voltage, and then it slowly falls back to the normal range. The amplitude of the output overshoot and the final recovery time are related to the equivalent impedance characteristics of the sensor, and the normal value of the output can be represented by its average value.

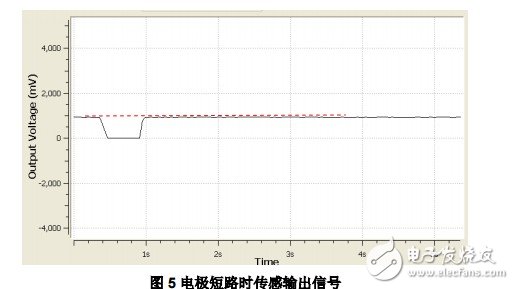
2.2 Short circuit of sensor electrode Short circuit of sensor electrode is a common type of installation error, and it will correspond to the fault state of electrode short circuit. Figure-5 shows the output after the sensor electrode is short-circuited and the fault detection is enabled, which is very similar to the output in normal application. The output voltage at this time also goes through three stages of drop, maintenance, and rise, but there is no overshoot phenomenon in the voltage after the rise.
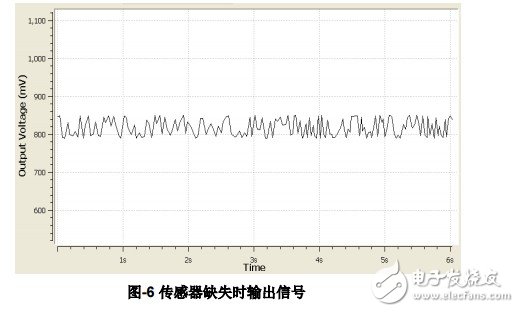
2.3 Missing sensor Missing or falling off sensor is another common electrode connection fault. Figure-6 and Figure-7 show the output voltage of the sensor before and after the fault detection function is enabled. Compared with the output during normal use, the output signal noise component is obvious after the sensor is missing, and the output voltage fluctuates greatly; after the fault detection is enabled, the sensor output characteristic does not change significantly and there is no voltage drop or climb. The degree of fluctuation of the output voltage can be assisted in the judgment with the help of the expected mean and mean square deviation of the sampled values.
3 Data analysis algorithm for electrode fault detection
The above test gives the corresponding output waveforms under common electrode failures of electrochemical sensors, and briefly analyzes the respective output characteristics. The following is a data analysis and processing flow for reference. The main implementation method is to traverse and search all the collected data after enabling the fault detection function, find out and store the characteristic information of its various changing edges, and distinguish the corresponding ones according to the characteristics of the jumping edge and whether there is overshoot. malfunction. When starting the fault detection, the microcontroller can modify the internal REFCN register of the LMP91000 through the I2C interface to realize the change of the sensor bias voltage. The duration of the bias voltage can be controlled by the timer; after the timing ends, the output data will be started. analysis.
Because the electrochemical sensor's complex physical and chemical properties make it sensitive to changes in bias voltage, the programmable feature of LMP91000 is used to generate artificial bias voltage disturbances to realize the detection of the electrode state. Considering that the gas concentration will not change rapidly in practical applications and cause the output to jump, which is different from the normal output signal, so that the electrode fault detection function can be quickly realized through the LMP91000. It should be emphasized that the amplitude and duration of the bias voltage change in fault detection should be strictly limited to prevent possible damage to the sensor itself. In addition, the fault detection of the three-electrode electrochemical sensor is very similar to that of the two-electrode, and the corresponding output characteristics are basically the same.
ConclusionThis is the end of the related introduction about lmp91000. Please correct me if there is any supplement.
Related reading recommendations: What is the structure and working principle of the electrochemical gas sensor?
A new rule from the Drug Enforcement Administration (DEA) threatens to upend the American hemp industry, and could even result in criminal prosecutions for manufacturers of CBD and delta-8 THC products.
The DEA says the [interim final rule," issued Aug. 20, is simply a matter of adjusting its own regulations to account for changes to the Controlled Substances Act that were mandated by the 2018 Farm Bill (or Agricultural Improvement Act) that legalized hemp and CBD production. The new rule [merely conforms DEA`s regulations to the statutory amendments to the CSA that have already taken effect," says the agency. The new rule doesn`t break any ground, according to the DEA.
But many experts on cannabis and hemp law say the DEA rule creates a potential pathway the law enforcement agency could use to prosecute hemp processors and producers of CBD (cannabidiol) and delta-8 THC (or Δ8THC) products. There are two issues: partially processed CBD, and [synthetically derived" delta-8 THC.
Cbd Pod System Oem,Cbd Vape Pod Oem,Best Cbd Pod System,Cbd Pod System
Shenzhen MASON VAP Technology Co., Ltd. , https://www.cbdvapefactory.com