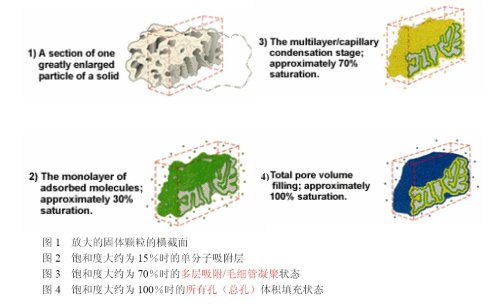
 Gas adsorption principle and process physical adsorption Physical adsorption is the most common type of adsorption. () The physically adsorbed molecules can move relatively freely on the surface of the sample. As more and more gas molecules are introduced into the system, the adsorbate molecules form a thin layer across the surface of the adsorbent. According to the well-known BET theory, assuming that the adsorbed molecules are monolayers, we can estimate the number of molecules required to cover the entire adsorbent surface, Nm (see Figure 2). The product of the number of adsorbed molecules Nm and the cross-sectional area of ​​the adsorbate molecules is the surface area of ​​the sample. Continued increase in the amount of gas molecules introduced will result in multiple layers of adsorption. The multilayer adsorption process and the capillary condensation process (see Figure 3) are performed simultaneously. The latter process can be fully described by the Kelvin equation. This equation quantifies the ratio of the residual (or equilibrium) gas pressure to the capillary size of the condensable gas. The aperture can be calculated from the equilibrium gas pressure using calculation methods such as Barrett, Joyner and Halenda (BJH). Therefore, we can make an experimental curve (ie isotherm) between the volume of the adsorbed gas and the relative saturation equilibrium pressure, and then convert it to obtain a cumulative or differential pore size distribution map. As the equilibrium adsorbate pressure tends to saturate, the pores are completely filled with adsorbate (see Figure 4). If you know the density of the adsorbate, you can calculate the volume it takes, and then calculate the total pore volume of the sample accordingly. If we reverse the adsorption process at this time, and gradually reduce the amount of gas from the system, the desorption isotherm can also be obtained. Adsorption and desorption isotherms rarely overlap due to differences in adsorption and desorption mechanisms. The hysteresis of the isotherms is related to the pore shape of the solid particles. Unlike physical adsorption, chemisorption is due to the formation of strong chemical bonds between the adsorbate molecules and specific locations on the surface, ie, chemically active sites. Therefore, the basic use of chemisorption is to calculate the number of surface active sites that may cause chemical and catalytic reactions.
Gas adsorption principle and process physical adsorption Physical adsorption is the most common type of adsorption. () The physically adsorbed molecules can move relatively freely on the surface of the sample. As more and more gas molecules are introduced into the system, the adsorbate molecules form a thin layer across the surface of the adsorbent. According to the well-known BET theory, assuming that the adsorbed molecules are monolayers, we can estimate the number of molecules required to cover the entire adsorbent surface, Nm (see Figure 2). The product of the number of adsorbed molecules Nm and the cross-sectional area of ​​the adsorbate molecules is the surface area of ​​the sample. Continued increase in the amount of gas molecules introduced will result in multiple layers of adsorption. The multilayer adsorption process and the capillary condensation process (see Figure 3) are performed simultaneously. The latter process can be fully described by the Kelvin equation. This equation quantifies the ratio of the residual (or equilibrium) gas pressure to the capillary size of the condensable gas. The aperture can be calculated from the equilibrium gas pressure using calculation methods such as Barrett, Joyner and Halenda (BJH). Therefore, we can make an experimental curve (ie isotherm) between the volume of the adsorbed gas and the relative saturation equilibrium pressure, and then convert it to obtain a cumulative or differential pore size distribution map. As the equilibrium adsorbate pressure tends to saturate, the pores are completely filled with adsorbate (see Figure 4). If you know the density of the adsorbate, you can calculate the volume it takes, and then calculate the total pore volume of the sample accordingly. If we reverse the adsorption process at this time, and gradually reduce the amount of gas from the system, the desorption isotherm can also be obtained. Adsorption and desorption isotherms rarely overlap due to differences in adsorption and desorption mechanisms. The hysteresis of the isotherms is related to the pore shape of the solid particles. Unlike physical adsorption, chemisorption is due to the formation of strong chemical bonds between the adsorbate molecules and specific locations on the surface, ie, chemically active sites. Therefore, the basic use of chemisorption is to calculate the number of surface active sites that may cause chemical and catalytic reactions. 
SP300VAC600W is a switching mode single-channel output high-precision AC power source, it`s cost effective and fully programmable for both basic frequency conversion and advanced AC power line or DC power disturbance test applications. This AC Power Supply adopts high speed DSP+CPLD control, high frequency PWM power technology and active PFC design to realize AC/DC stable output.
APM ac output power supply is featured with high power density, high reliability and high precision, meanwhile it possesses operation interface of touch screen and keys manually. It is able to analog output normal or abnormal input for electrical device to meet test requirements.
Some features as below:
- 4.3"large touch color screen
- AC+DC mixed or independent output mode
- Capable of setting output slope/phase angle
- Built-in IEC standard test function
- Built-in multiple protections
- Built-in power meter
- Support impedance function
- Support for LIST/PULSE/STEP mode & Transient mode
- Standard RS232/RS485/USB, Optional GPIB//LAN
- Support harmonics/inter-harmonics simulation and measuring function
- Support for USB data import/export and scree nap from front panel
600W AC Power Supply,Variable AC Power Supply,Portable Ac Power Source,Ac Voltage Source
APM Technologies Ltd , https://www.apmpowersupply.com